
Sodium acetate hydrate, Puratronic™, 99.9985% (metals basis), Thermo Scientific Chemicals | Fisher Scientific
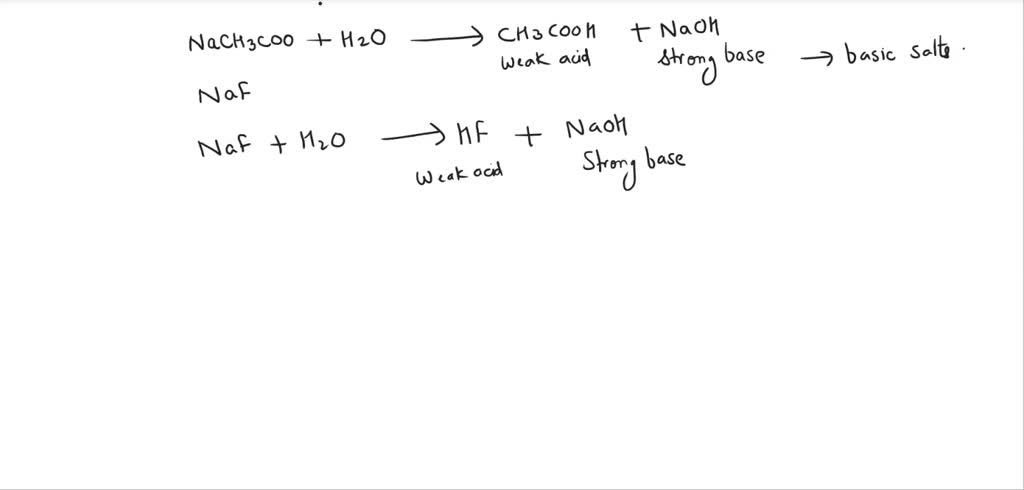
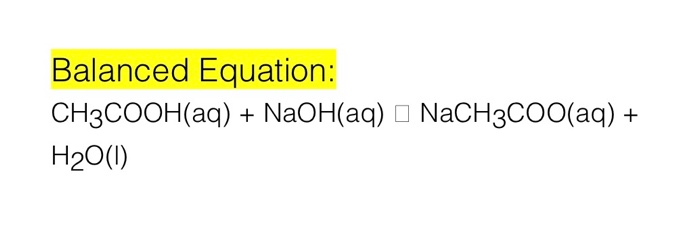
SOLVED: Write chemical equations to show that NaCH3COO and NaF are basic salts. Indicate which of the two salts are more basic in aqueous solutions, and explain why.

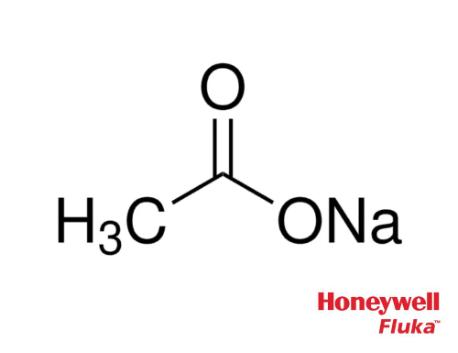
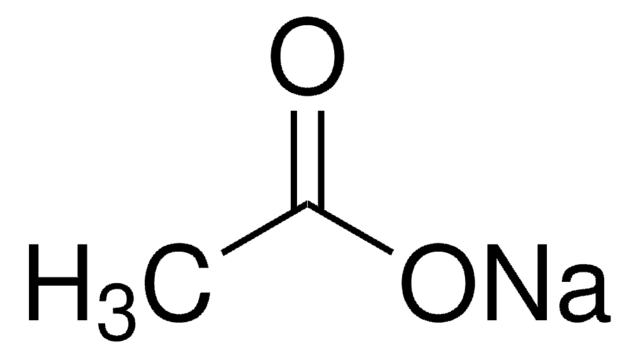
Sodium Acetate(CH3COONa) - Structure, Properties, Preparations, Uses, Important questions, FAQs of sodium acetate.

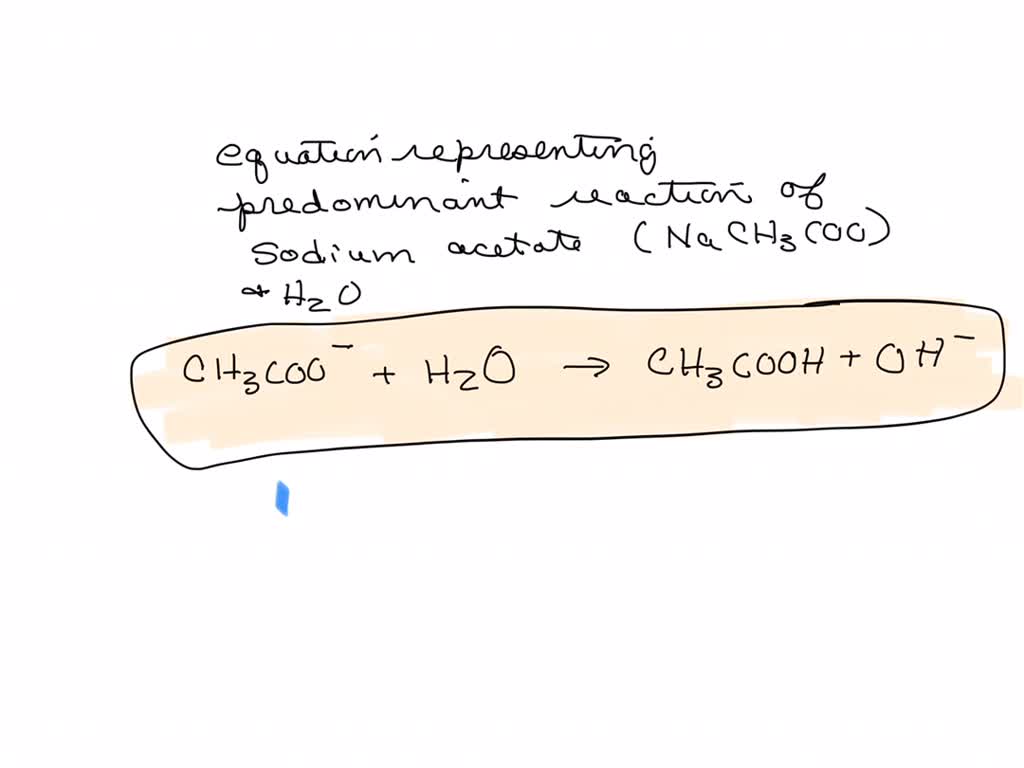
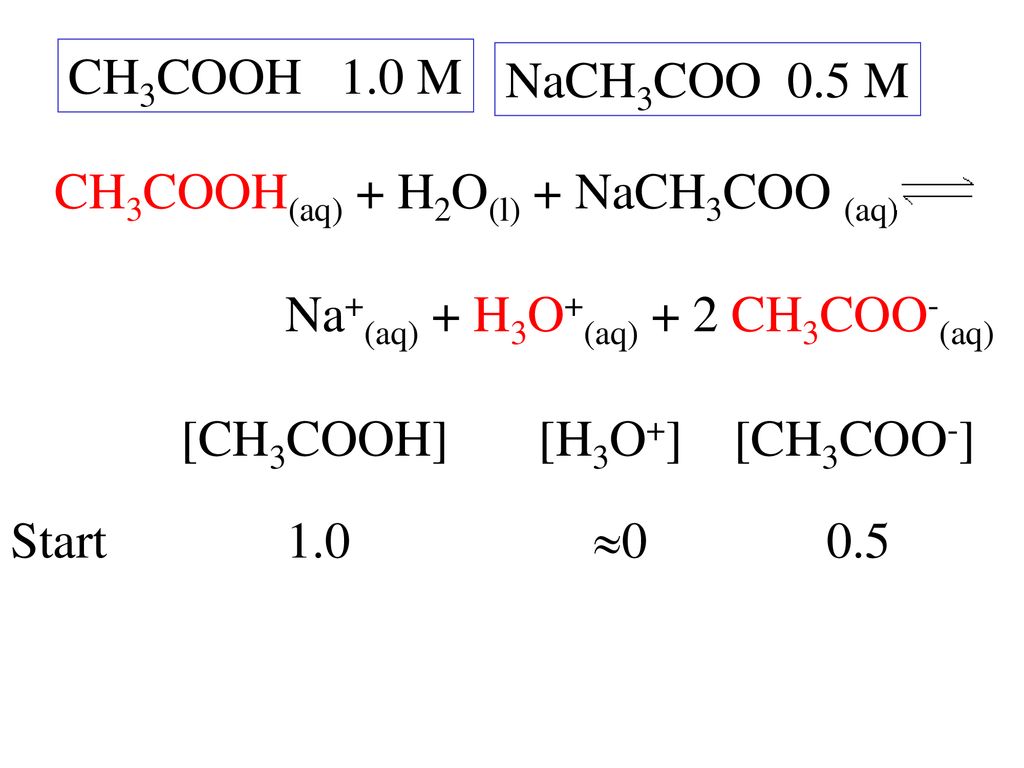
For sodium acetate solution in water, the given equilibrium reaction occur:CH 3 COO aq + H 2 O l hydrolysis ⇌ CH 3 COOH aq + OH aqWhich of the following describes



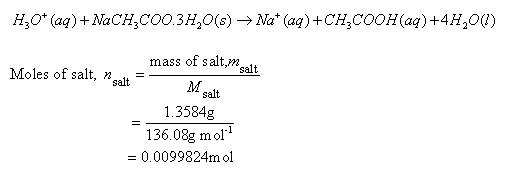




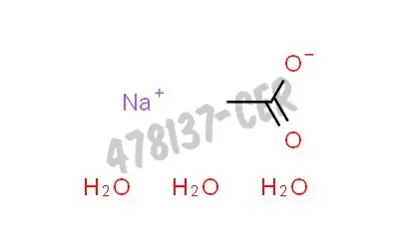


![Sodium Acetate Trihydrate [CH3COONa.3H2O] Molecular Weight Calculation - Laboratory Notes Sodium Acetate Trihydrate [CH3COONa.3H2O] Molecular Weight Calculation - Laboratory Notes](https://www.laboratorynotes.com/wp-content/uploads/2022/12/sodium-acetate-trihydrate-molecular-weight-calculation-300x228.jpg)
