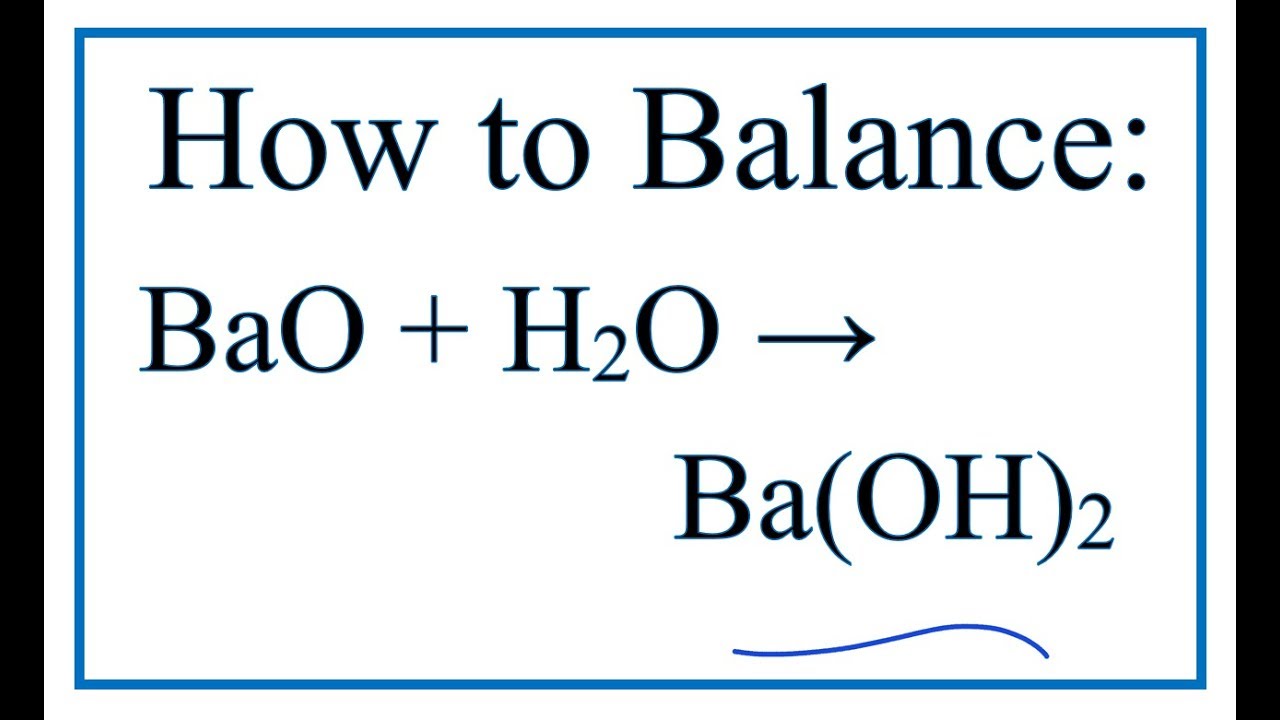
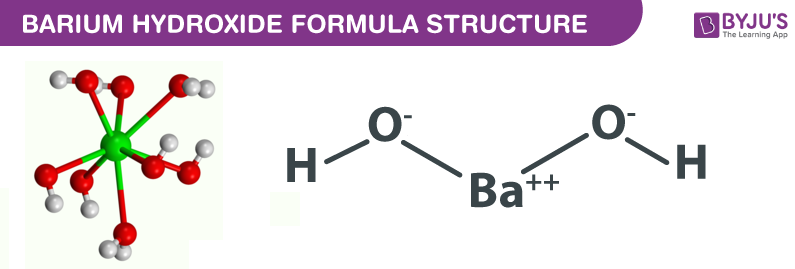
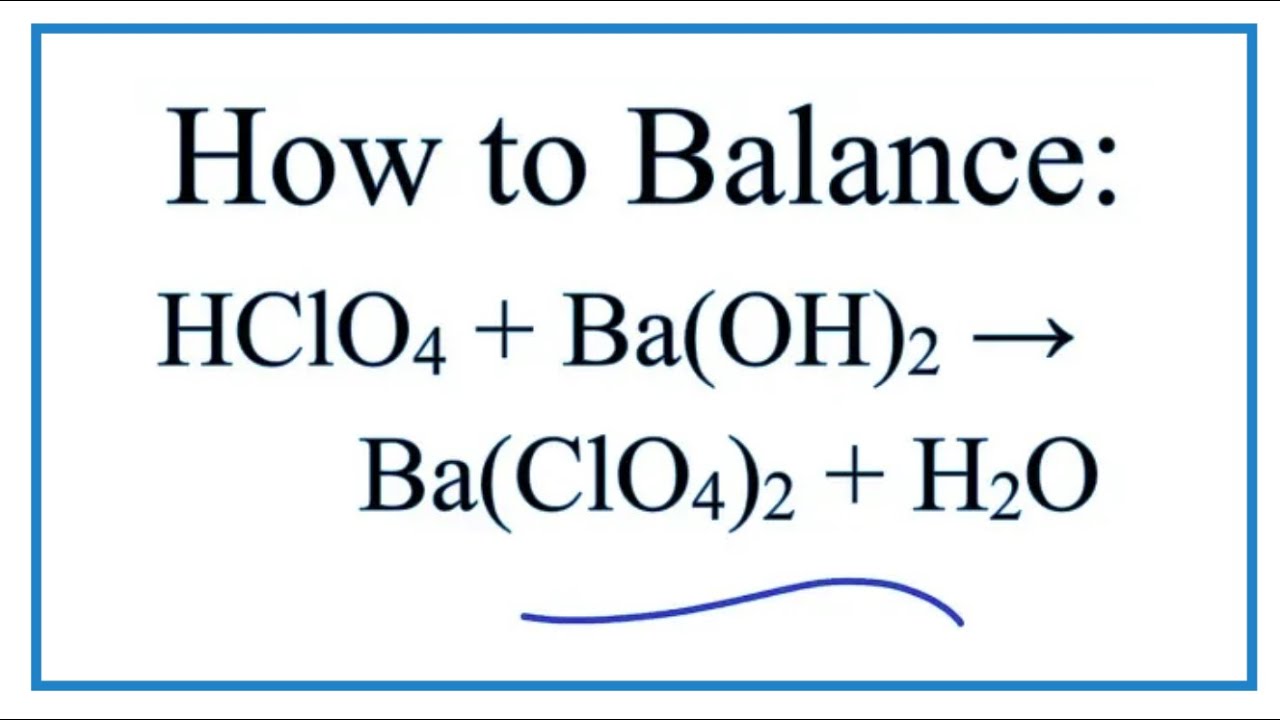
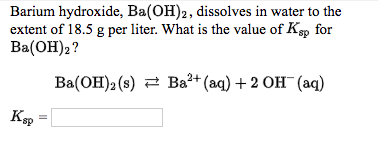BALANCE THIS GIVEN EQUATION BY THE HELP OF THE ALTERNATE WAY OF BALANCING NOT THE TRADITIONAL WAY BY SHOWING THE APPROPRIATE STEPS ELABORATELY. EQN : Ba(OH)2 + NH4Cl —> BaCl2 + NH3 + H2O

Question Video: Determining the Products of the Neutralization Reaction of Barium Hydroxide Ba(OH)₂ with Carbonic Acid H₂CO₃ | Nagwa

Chine baryum hydroxyde poudre fabricants, fournisseurs et usine - produits - Yingfengyuan Industrial Group Limited

Barium Hydroxide Monohydrate Ba (OH) 2. H2O - China Barium Hydroxide, Barium Hydroxide Monohydrate | Made-in-China.com

SOLVED: Consider the acid-base reaction: 2 HNO2 (aq) + Ba(OH)2 (aq) â†' Ba(NO2)2 (aq) + 2 H2O Predict the products of the acid-base reaction: Ba2+ (aq) + 2 NO2- (aq) + 2
Acheter de l'hydroxyde de baryum? - Hydroxyde de baryum de la meilleure qualité à un prix avantageux chez Laboratoriumdiscounter